Yeah, my point is principally that the "dissolving" analogy is just plain wrong. Vapor is not "dissolved" in air, it is part of the mix of gases, they all have the same mean displacement lenght, and they only interact by perfectly elastic collisions.photofly wrote:I believe it's hard to predict the density of liquid solutions from the density of the solvent and the density of the solute; in liquids the interactions between molecules is important. I think your valid point is that in gas-in-gas solutions, it isn't.If the moisture in the air was analog to salt being dissolved into water (which is the analogy most people have in mind when they think about air being saturated or warm air "holding" more moisture), the moist air would be DENSER then dry air,
How does falling rain effect relative humidity?
Moderators: sky's the limit, sepia, Sulako, Right Seat Captain, lilfssister, North Shore
Re: How does falling rain effect relative humidity?
Re: How does falling rain effect relative humidity?
So I dig up Met Theory (Section 1, Aerocourse 5th ed) and re-read the learning question ... RELATIVE HUMIDITY MAY BE DEFINED AS: c) THE AMOUNT OF MOISTURE IN THE AIR COMPARED TO THE AMOUNT THE AIR COULD HOLD AT THAT TEMPERATURE AND PRESSURE.
Here's an instance where it's not necessarily to push you to the idea the air is "holding" the vapour, just using fewest words to introduce the concept of RH in a practical way.
The very first question: "The amount of water vapour that the air can hold is determined by its: b) temperature".
It steers to thinking about it as a certain dimension (an area of air) containing/sustaining/having a certain amount of moisture in it, represented by %RH.
Hold: to be filled by; contain. (is one of many definitions listed for the word, The Free Online Dictionary/Thesaurus)
Then "could hold"/"can hold" works OK there in those questions. Yet saying 'air is "holding" water vapour' (loosely stated) admittedly becomes too much of a stretch (FALSE), as you have explained above.
Here's an instance where it's not necessarily to push you to the idea the air is "holding" the vapour, just using fewest words to introduce the concept of RH in a practical way.
The very first question: "The amount of water vapour that the air can hold is determined by its: b) temperature".
It steers to thinking about it as a certain dimension (an area of air) containing/sustaining/having a certain amount of moisture in it, represented by %RH.
Hold: to be filled by; contain. (is one of many definitions listed for the word, The Free Online Dictionary/Thesaurus)
Then "could hold"/"can hold" works OK there in those questions. Yet saying 'air is "holding" water vapour' (loosely stated) admittedly becomes too much of a stretch (FALSE), as you have explained above.
Re: How does falling rain effect relative humidity?
I would try as much as possible to not use aviation books as a reference when it comes to meteorology. I think photofly would say pretty much the same thing when it comes to aerodynamics.
Introducing concepts in a practical way doesn't mean you should use wrong analogies and botch the explanations.
Air does not hold vapor, it is not holding vapor... Say it however you like, it won't be correct.
In the atmosphere, air is not even "filled" by vapor. Not more that you would say that nitrogen is filled by oxygen.
I don't have a problem with simplifications when they don't lead to misunderstandings and endless mistakes. For example I don't care that the general convention in electricity is that current flows from positive to negative, while we know that it's in fact the opposite that occurs. In such case, this mistake has no bearing on the general understanding when studying circuits and leads to no "intuitive mistakes". Saying that air holds moisture does.
Introducing concepts in a practical way doesn't mean you should use wrong analogies and botch the explanations.
Air does not hold vapor, it is not holding vapor... Say it however you like, it won't be correct.
In the atmosphere, air is not even "filled" by vapor. Not more that you would say that nitrogen is filled by oxygen.
I don't have a problem with simplifications when they don't lead to misunderstandings and endless mistakes. For example I don't care that the general convention in electricity is that current flows from positive to negative, while we know that it's in fact the opposite that occurs. In such case, this mistake has no bearing on the general understanding when studying circuits and leads to no "intuitive mistakes". Saying that air holds moisture does.
Re: How does falling rain effect relative humidity?
I think it's a fair point. It's not the air that's holding the water vapour. It's the empty space that's holding the water vapour, just like it's holding oxygen and nitrogen, a bit of argon, some ppm of carbon dioxide etc.
Re: How does falling rain effect relative humidity?
And there is so much space in between all of the molecules that pressure (unless it is ridiculously high) is not a factor influencing the amount of water that can evaporate.
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
What I'm suggesting is 100% is maximum.
The problem being that conditions can change how much maximum is.
The maximum might be 10 units. Then the air warms up and the maximum is 20 units. Now the same amount of water 10 units, that was 100% is 50%. The 20 units will be 100%. There is never 200%.
The problem being that conditions can change how much maximum is.
The maximum might be 10 units. Then the air warms up and the maximum is 20 units. Now the same amount of water 10 units, that was 100% is 50%. The 20 units will be 100%. There is never 200%.
Re: How does falling rain effect relative humidity?
Now cool the air again. The 100% can't condense because it doesn't have any nucleation sites, and is now at a rh of 200%. You have achieved supersaturation.The 20 units will be 100%. There is never 200%.
Some time later, introduce some dust, or some potassium iodide crystals, and <boof> some moisture condenses instantly - instant cloud - leaving "only" 100% rh.
It's very similar to the way you can cool liquid water way way below 0 degrees C without it freezing, if you keep it clean and still. Disturb it - and suddenly it will freeze solid almost instantly.
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Re: How does falling rain effect relative humidity?
Lol.
After cooling, the atmosphere in the box contains still 20 units of water vapour because it isn't able to condense. But at the new lower temperature, just 10 is enough to saturate(*). So the rh is 200%.
(*) which means that at equilibrium the space in the box can't hold more than 10 units of water vapour. They key is the "equilibrium" bit. When it holds 20 units it's not at equilibrium.
After cooling, the atmosphere in the box contains still 20 units of water vapour because it isn't able to condense. But at the new lower temperature, just 10 is enough to saturate(*). So the rh is 200%.
(*) which means that at equilibrium the space in the box can't hold more than 10 units of water vapour. They key is the "equilibrium" bit. When it holds 20 units it's not at equilibrium.
Re:
Since you're a funny guy, I'm not sure if you are really confused or trolling here...Beefitarian wrote:What I'm suggesting is 100% is maximum.
Please re-read what was posted previously in this thread (the links also). You don't understand the definition of RH.
Would you say that the price of a good could not rise by 150%, since 100% is always the maximum of something?
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Little of both of course. You have found one of my special pet peeves. The concept of not worrying about 100 being all the percent is typically used for tricking you into investing your money with me, so I can promise to put in 110% effort to increase your investment by at least 300% taking my 20% followed by you losing 30% and having 45% of the money you deposited left in you account.
Keep going I'm starting to learn. Maybe I can eventually take the CPL written. I'd like to score a couple hundred percent.
How does the increased temp change things to allow the extra units?
Keep going I'm starting to learn. Maybe I can eventually take the CPL written. I'd like to score a couple hundred percent.
How does the increased temp change things to allow the extra units?
Re: How does falling rain effect relative humidity?
We can just say AIRSPACE and make all the aviators happy.photofly wrote:It's not the air that's holding the water vapour. It's the empty space that's holding the water vapour.
Last edited by pdw on Tue Apr 23, 2013 7:16 pm, edited 1 time in total.
Re: How does falling rain effect relative humidity?
Good question. I don't know the exact physics.How does the increased temp change things to allow the extra units?
Generally speaking, increased temperature means the range of kinetic energies of the molecules is higher. At a given temperature some molecules already condensed in droplets pick up enough energy through collisions to break free of the forces (Van Der Waals forces) that attract them to other molecules. In water, those are hydrogen bonds, which are attractive forces caused between the hydrogen atom of one molecule - which looks a lot like a "naked" proton, once the atom's single electron is bound in a bonding orbital with the oxygen. The strong electric field from the hydrogen's single nuclear proton polarizes the electron cloud surrounding the oxygen atom of other water molecules and is then attracted by the dipole field it has created. That means that liquid water molecules stick together in clumps of several hundred molcules' size, someone worked out. To break free of those forces and float off as a gaseous water molecule takes a lot of energy, so only those molecules that are high up in the distribution of kinetic energies can do it. If you increase the "temperature" of the water by some amount, more water molecules have sufficient energy to break out into gaseous phase.
Going the other way, gas phase water molecules that collide and don't between them have enough energy to bounce apart will stick together. But that's where the physics is tricky, because there's some energy barrier to forming new droplets which is lowered by nucleation sites like dust grains.
The whole subject of phase transitions (liquid to/from gas, or solid to/from liquid, as well as other more esoteric ones like phase changes in solids) is a big topic in physics.
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Re: How does falling rain effect relative humidity?
You might not know the exact physics (heck, who does?), but it seems good enough here!photofly wrote:Good question. I don't know the exact physics.How does the increased temp change things to allow the extra units?
Beef, what photofly said is basically the following: higher temperature means more kinetic energy per molecule (they move more). When stuff moves a lot, it collides a lot, and wants to get the hell out of this place. Why stay close together (solid or liquid) when you move so much your neighbors can't stand you anymore? Might as well leave and become freaking gas, flying alone very fast all by yourself like a boss. So on average, the higher the temperature, the more energetic molecules you have, so there are more that prefer the freedom of open space instead of a liquid or solid prison.
Re:
Took me a while to work out how the Colonel can set his L39 to 105% N1, until I realised 100% is really just an arbitrary number. Kind of a Spinal Tap thingBeefitarian wrote:Little of both of course. You have found one of my special pet peeves. The concept of not worrying about 100 being all the percent is typically used for tricking you into investing your money with me, so I can promise to put in 110% effort to increase your investment by at least 300% taking my 20% followed by you losing 30% and having 45% of the money you deposited left in you account.
Keep going I'm starting to learn. Maybe I can eventually take the CPL written. I'd like to score a couple hundred percent.
How does the increased temp change things to allow the extra units?

- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Choosing percentage to arbitrary numbers to represent something that would be slightly more difficult to explain using the term properly should not bother me so much but it does.
It's like Internet things people don't understand. +1 means you agree and want to add your vote +10 means you don't understand what +1 is.
Being picky FTW.
Edit: I fully understand this is hypocritical of me since my spelling is so bad. Sorry.
It's like Internet things people don't understand. +1 means you agree and want to add your vote +10 means you don't understand what +1 is.
Being picky FTW.
Edit: I fully understand this is hypocritical of me since my spelling is so bad. Sorry.
Re: How does falling rain effect relative humidity?
100% RH really means something, it's not abritrary. It means the EQUILIBRIUM over a FLAT SURFACE of PURE water is reached at a given temperature. It does not mean it's the maximum amount of vapor there can be in a given volume at a certain time.Beefitarian wrote:Choosing percentage to arbitrary numbers to represent something that would be slightly more difficult to explain using the term properly should not bother me so much but it does.
Yes indeed.Beefitarian wrote:Being picky FTW.
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Re: How does falling rain effect relative humidity?
The water vapour is clear/ invisible, becoming visible cloud only as it condenses on condensation nuclei [or if "super saturated" air-space exists (which would be far beyond the amount of a normal "equillibrium" H20 content, 100%RH)] ... then followed by "a cascade of collisions and coalescence" that turns the tiny 'condensates' into sizeable raindrops.trampbike wrote:(with the help of our little fellas condensation nucleis and after a cascade of collisions and coalescence)
Re:
First, forget the air. As far as saturation and condensation go, pretty much the same process would happen with only vapor and conensation nucleis (CN). Vapor doesn't care too much about oxygen or nitrogen, and neither do they about vapor.Beefitarian wrote:Let's get into some cloud talk here.
Clouds form before equilibrium. Are they vapor that is not attaching molecularly to the air yet suspended in it or does that factor in?
I know this will get intensely complicated here. Bear with me zoning in and out of logic.
Clouds can form before, after or at equilibrium, it really depends on the amount and type of CN that are hanging around.
Clouds are not made of vapor, they are made of water or ice. The only difference between raindrops and cloud droplets is size:
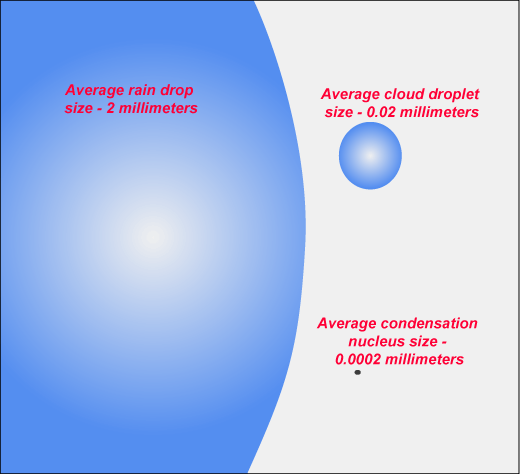
The tiny droplets that are constituting clouds are so light compared to their surface (the smaller something is, the bigger its surface/mass is for a given shape) that gravity has almost no effect on them compared to friction from the generally lightly ascending air found in clouds. That's why they don't fall. They have to grow much larger before they become raindrops and be able to... you guessed: DROP!
- Beefitarian
- Top Poster

- Posts: 6610
- Joined: Wed Dec 01, 2010 10:53 am
- Location: A couple of meters away from others.
Ok good, I think I know a small amount about the cloud versus vapor here.
Vapor is water molocules in the form of gas that do not attatch to the air, instead they occupy the space between the other gasses of an air mass. Gas is a happy invisible thing that floats around as part of our atmosphere. Pilots, bugs, fish, squirrels and birds get to fly in it.
A certain amount of water molecules can fit in the space between a certain amount of what ever content is making up a particular air mass. Depending on the pressure and temperature conditions, the space will change and can allow more vapor in or just a gathering of the present vapor to the center of the airmass. Once all the molecules are present in balanced amounts that is equalibrium. If the temperature rises or pressure lowers there will be more space allowing more vapor to gather until a new equalibrium is achieved.
If the temperature lowers or the pressure increases the water molecules have less space and want to condensate. This is where we diverge slightly, because that amount of vapor may be more than should accupy the particular space. Then some refer to that amount as being more than 100% relative humidity. There is extra units of water vapor that snuck in when there was more space for them but now they can't get out.
Condensation is the vapor changing from gas to liquid. The water vapor cannot do that unless there is a surface to attatch to so it can become liquid again. At night plant surfaces cool off and water condensates on to them, dew drops. If there is dust floating in the atmosphere, which there usually is the water can condensate onto that and it's called a "Condensation Nucleus."
Now we have a very small partical of water liquid attatched (not chemically just physically) to a very small partical of solid light enough to continue floating, because it's visible we see it and call a large gathering of them a cloud. More vapor can attatch to each liquid water particle until it becomes too heavy to stay airborne at which point it falls as a rain drop. This condensation releases the energy that was causing the liquid to be gas, we call that "Latent Heat." As the air is heated it will rise and cool causing motion in the cloud.
If the rain drop freezes, then it will become an ice pellet or snow flake.
Vapor is water molocules in the form of gas that do not attatch to the air, instead they occupy the space between the other gasses of an air mass. Gas is a happy invisible thing that floats around as part of our atmosphere. Pilots, bugs, fish, squirrels and birds get to fly in it.
A certain amount of water molecules can fit in the space between a certain amount of what ever content is making up a particular air mass. Depending on the pressure and temperature conditions, the space will change and can allow more vapor in or just a gathering of the present vapor to the center of the airmass. Once all the molecules are present in balanced amounts that is equalibrium. If the temperature rises or pressure lowers there will be more space allowing more vapor to gather until a new equalibrium is achieved.
If the temperature lowers or the pressure increases the water molecules have less space and want to condensate. This is where we diverge slightly, because that amount of vapor may be more than should accupy the particular space. Then some refer to that amount as being more than 100% relative humidity. There is extra units of water vapor that snuck in when there was more space for them but now they can't get out.
Condensation is the vapor changing from gas to liquid. The water vapor cannot do that unless there is a surface to attatch to so it can become liquid again. At night plant surfaces cool off and water condensates on to them, dew drops. If there is dust floating in the atmosphere, which there usually is the water can condensate onto that and it's called a "Condensation Nucleus."
Now we have a very small partical of water liquid attatched (not chemically just physically) to a very small partical of solid light enough to continue floating, because it's visible we see it and call a large gathering of them a cloud. More vapor can attatch to each liquid water particle until it becomes too heavy to stay airborne at which point it falls as a rain drop. This condensation releases the energy that was causing the liquid to be gas, we call that "Latent Heat." As the air is heated it will rise and cool causing motion in the cloud.
If the rain drop freezes, then it will become an ice pellet or snow flake.
Re: How does falling rain effect relative humidity?
You're getting closer, but you have to throw out the concept of "space in between" molecules. In a gas, there is plenty of space, more than you can imagine. Pressure plays no role in the condensation/evaporation process (perfect gas approximation is good for plenty of atm of pressure!). What is important is the average KINETIC ENERGY of the molecules: temperature. When they have a lot of it, the molecules break their bonds and become lonely riders (gas), when they have less, they become liquid or solid. That's it.
In bold is where you are wrong, what is not in bold kind of makes sense!
In bold is where you are wrong, what is not in bold kind of makes sense!
Beefitarian wrote:Ok good, I think I know a small amount about the cloud versus vapor here.
Vapor is water molocules in the form of gas that do not attatch to the air, instead they occupy the space between the other gasses of an air mass. Gas is a happy invisible thing that floats around as part of our atmosphere. Pilots, bugs, fish, squirrels and birds get to fly in it.
A certain amount of water molecules can fit in the space between a certain amount of what ever content is making up a particular air mass. Depending on the pressure and temperature conditions, the space will change and can allow more vapor in or just a gathering of the present vapor to the center of the airmass. Once all the molecules are present in balanced amounts that is equalibrium. If the temperature rises or pressure lowers there will be more space allowing more vapor to gather until a new equalibrium is achieved.
If the temperature lowers or the pressure increases the water molecules have less space and want to condensate. This is where we diverge slightly, because that amount of vapor may be more than should accupy the particular space. Then some refer to that amount as being more than 100% relative humidity. There is extra units of water vapor that snuck in when there was more space for them but now they can't get out.
Condensation is the vapor changing from gas to liquid. The water vapor cannot do that unless there is a surface to attatch to so it can become liquid again. At night plant surfaces cool off and water condensates on to them, dew drops. If there is dust floating in the atmosphere, which there usually is the water can condensate onto that and it's called a "Condensation Nucleus."
Now we have a very small partical of water liquid attatched (not chemically just physically) to a very small partical of solid light enough to continue floating, because it's visible we see it and call a large gathering of them a cloud. More vapor can attatch to each liquid water particle until it becomes too heavy to stay airborne at which point it falls as a rain drop. This condensation releases the energy that was causing the liquid to be gas, we call that "Latent Heat." As the air is heated it will rise and cool causing motion in the cloud.
If the rain drop freezes, then it will become an ice pellet or snow flake.
Re: How does falling rain effect relative humidity?
EDITs ... "pressure" changes the air density too, yes, ... as does "less space"/"more space": .......colder/warmer; but the change in "space 'between' " (as shown in trampbike's previous posts), as far as room for the water vapour is concerned, really makes 'no difference' ('stays same') as the moisture holding capacity of the airspace isn't affected significantly whether warm or cold air.
the "snow flake" is sublimation
the "snow flake" is sublimation
Last edited by pdw on Sat Apr 27, 2013 4:54 am, edited 3 times in total.
Re: How does falling rain effect relative humidity?
Wrong. Why do you think cold air is denser than warm air?pdw wrote: "Less space"/"more space": Space 'between' stays same despite being cooler/warmer, it's just that slowed molecules (cooler) collide less.
As the temperature rises, the molecules have higher kinetic energy, and the average distance in between them increases.


